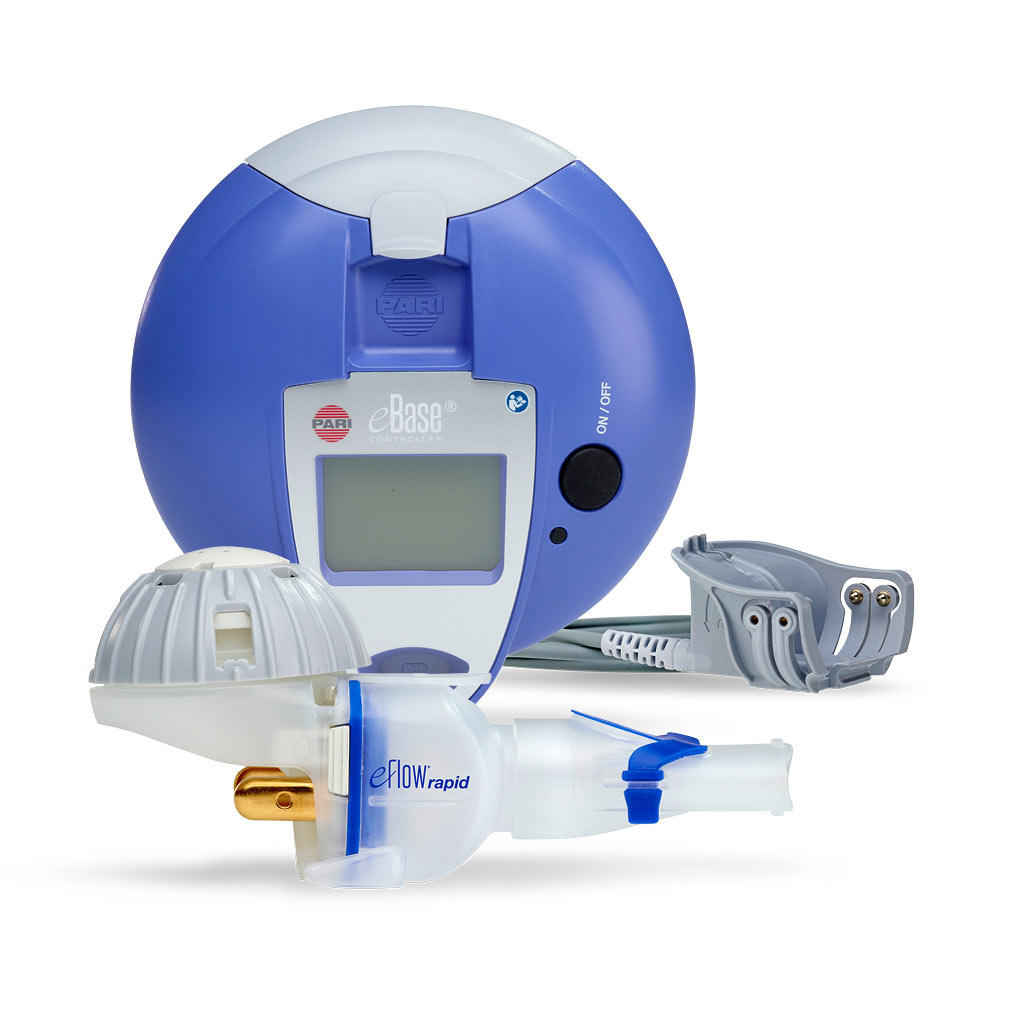
Magnair™ an eFlow® Closed System Nebulizer together with Sunovion's Lonhala™ is the first eFlow technology based product to receive FDA Approval to Treat Chronic Obstructive Pulmonary Disease (COPD)
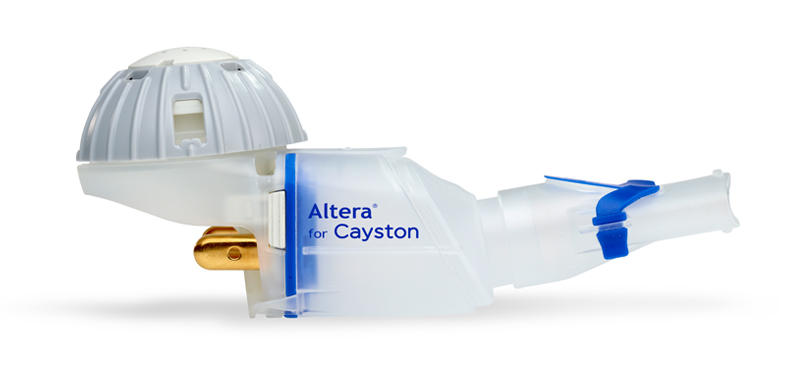
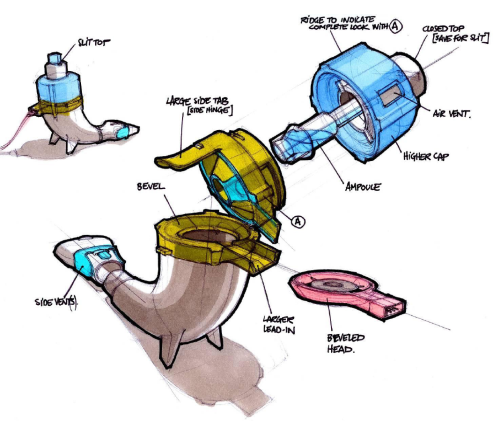
FDA-Approved Patient Labeling Patient Information CAYSTON® (kay-stun) (aztreonam for inhalation solution) Read this Patient Inf